- Concentration determination: spectrophotometry
- Endotoxin level determination: LAL method
- Drug to antibody ratio (DAR) determination: LC-MS, HPLC-HIC, RP-HPLC, etc.
- Purity determination: SEC/ RP/ HIC-HPLC, SDS-PAGE, etc.
- Residues of free drug determination: LC-MS/MS
- Residues of solvent determination: GC
- Site mapping determination: LC-MS
- Sterility test: membrane filtration/direct inoculation
ADC Binding Properties
- Biacore 8K
- Octet Red
- Biotek Plate Reader
- FACS Canto II
Physiochemical Properties
- Biomolecule/Linker-Payload Residue LC-MS
- SEC/HIC/RP/CEX-HPLC
- SDS-PAGE
- CE-SDS/iCIEF
ADC Stability Studies
- Uncle Protein Stability
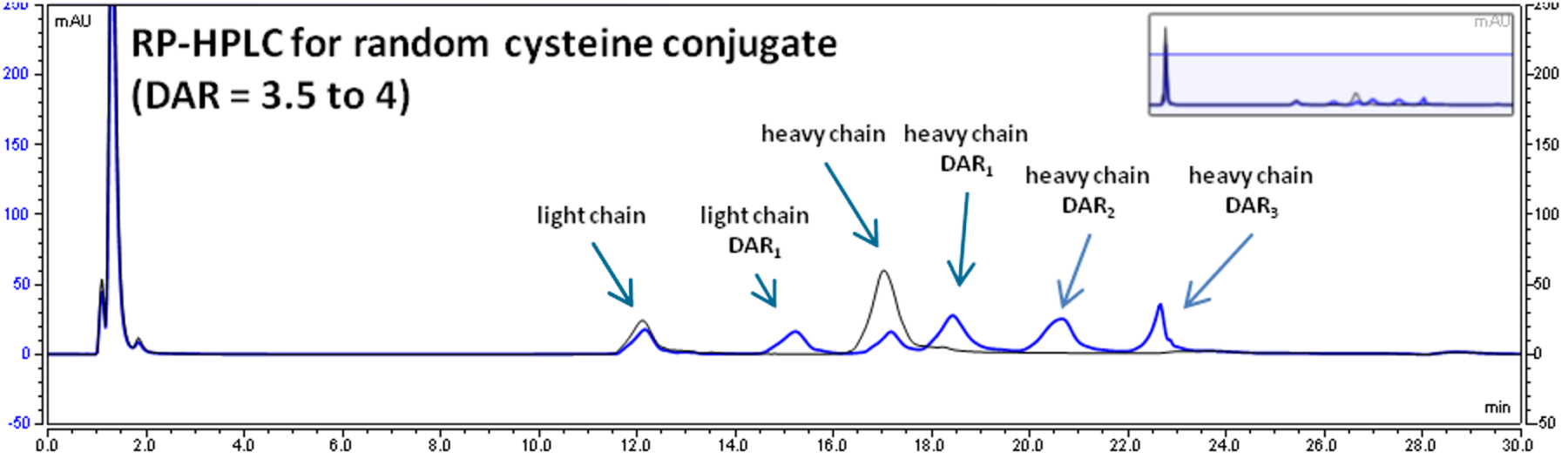
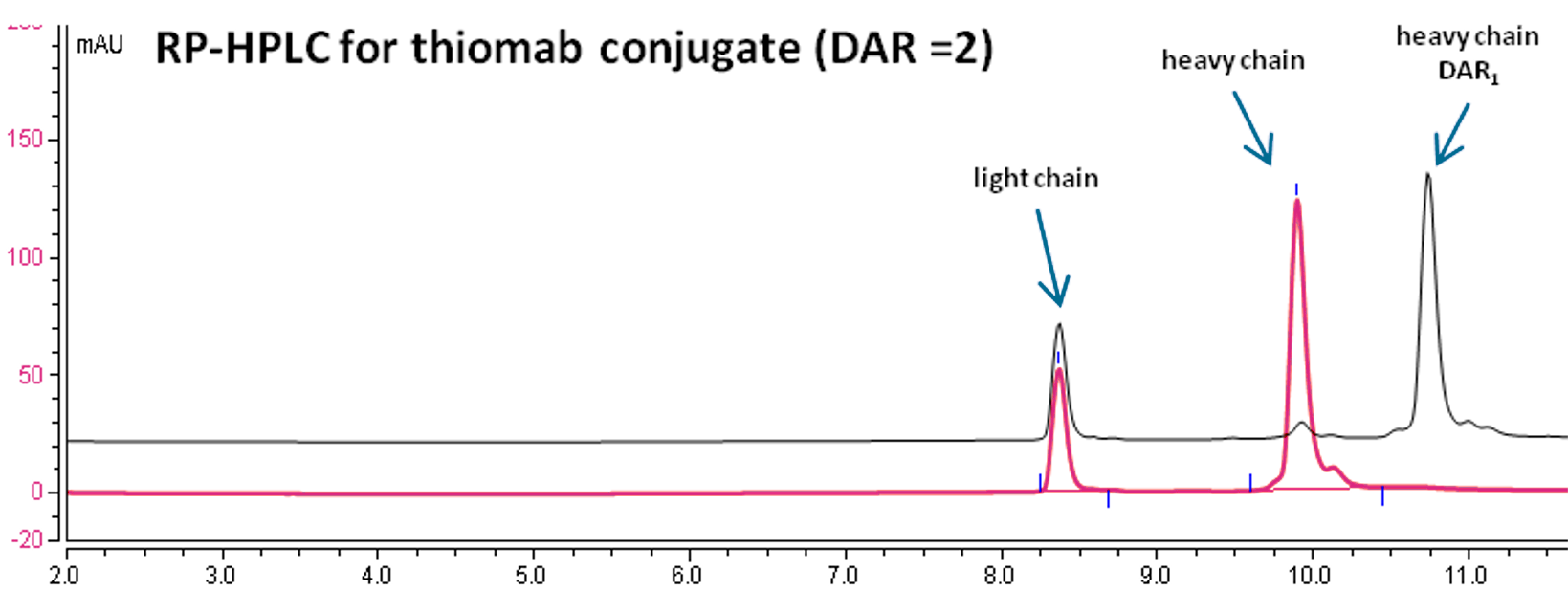
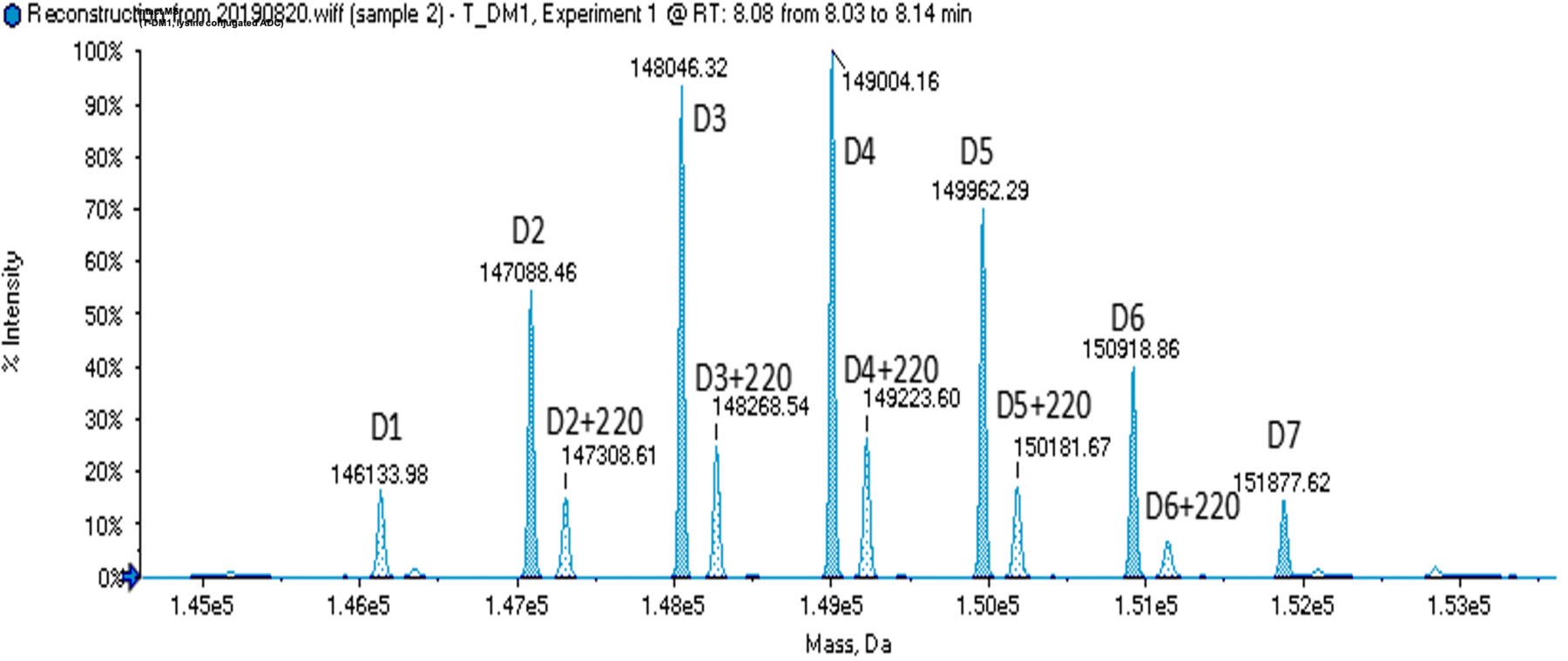
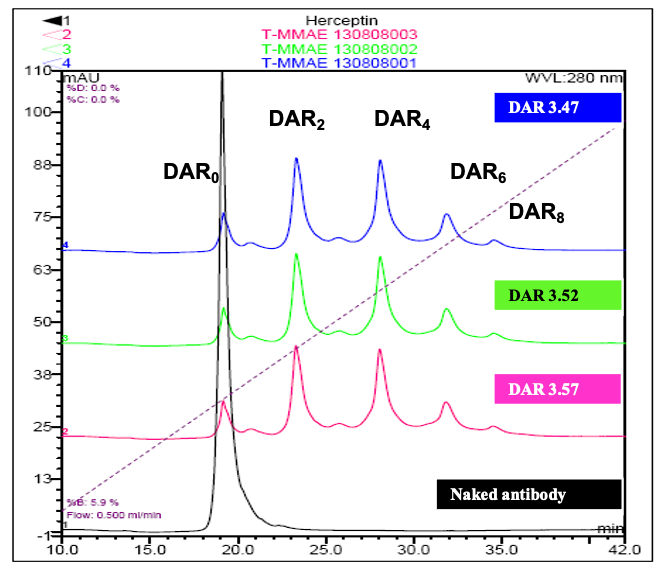
DAR determination by multiple methodologies
LC-MS
HPLC-HIC
Reverse Phase-HPLC