- Mouse, rat, monkey SC, IP, IV bolus, IV infusion
- Bio-analytical development
- PK/PD modeling and simulation
- In vitro stability in biological matrices
- Single dose, repeated dose, dose range finding in rats and monkeys
- Efficacy studies
- 160+ cell line xenografts and 270+ patient derived xenografts
- Biodistribution
- Imaging with labeled mAb or ADC
- Ex vivo tissue concentration analysis
In Vivo Efficacy Study with Target Positive Xenograft Model
Tumor Volume Changes in SCID Mice Bearing NCI-N87 Xenografts
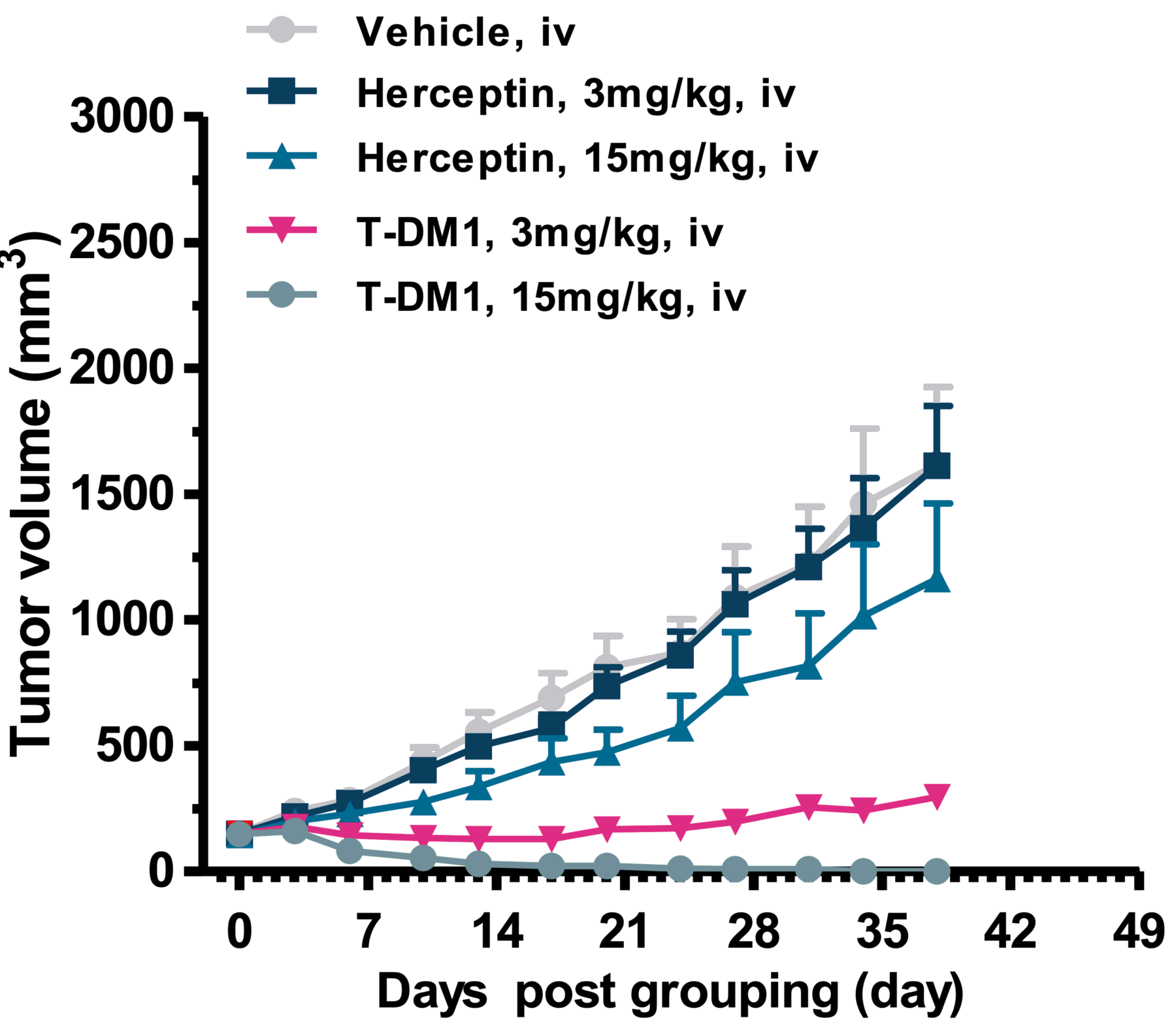
In-house prepared Trastuzumab-DM1 was tested in comparison with trastuzumab (Herceptin) in an in vivo study using gastric tumor cell Line NCI-N87 xenograft model of NOD-SCID mice. The tumor growth curves demonstrate tumor growth inhibition and complete regression after 3 doses i.v. of T-DM1 (3mg/kg and 15 mg/kg) compared to insignificant efficacy of naked antibody (n = 10 per group).
